Parkinson’s research is developing rapidly. While disease-modifying therapies are still in development, improved daily symptom control remains of central importance for people living with Parkinson’s today.
In this second article of our three-part series on new Parkinson’s therapies, we turn our attention to next-generation dopamine treatments. The focus is on tavapadone – a smarter, more selective dopamine therapy designed to alleviate motor symptoms while avoiding many of the side effects of current medications. This oral therapy is currently in late-stage clinical development for both early and advanced stages of Parkinson’s disease and represents an important advance in symptomatic treatment.
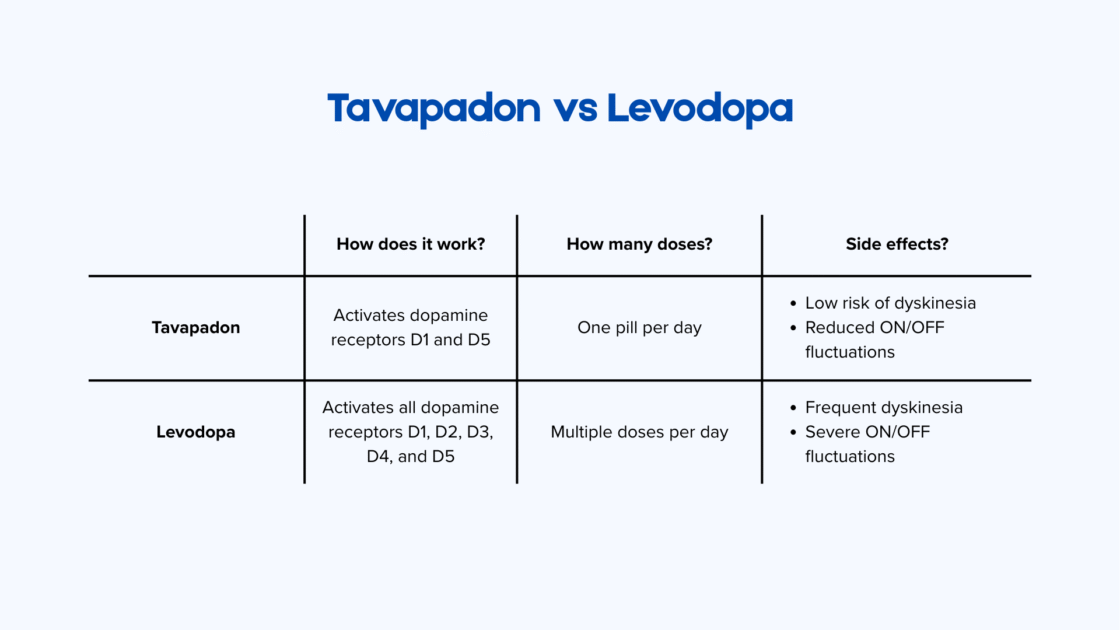
How does tavapadone differ from levodopa in Parkinson’s disease?
There are five dopamine receptors in the brain: D1, D2, D3, D4 and D5. The D1 and D5 receptors play a central role in motor control, while D2, D3 and D4 are more involved in neuron development. When the body converts levodopa into dopamine, dopamine activates all five receptors simultaneously to restore motor function. This overstimulation contributes to the development of dyskinesia.
Tavapadon is a dopamine therapy developed by the pharmaceutical company AbbVie (USA). It is highly selective and activates only the D1 and D5 receptors. This targeted approach can help to avoid side effects associated with broader activation of dopamine receptors. In addition, tavapadon remains active in the body for 24 hours, allowing it to be taken once daily 1.
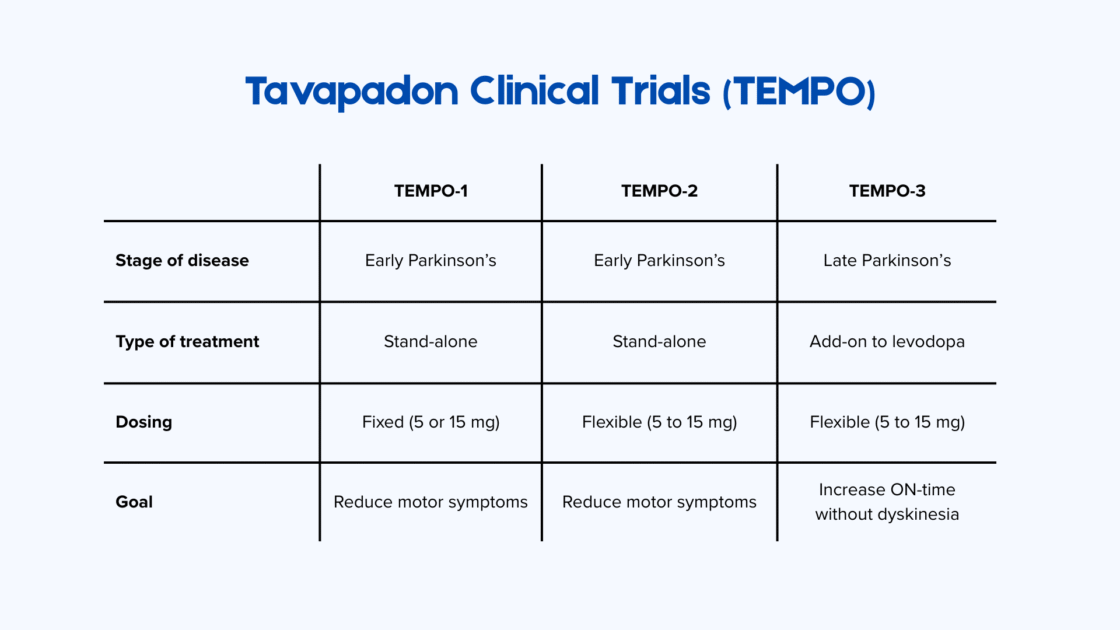
Phase 3 TEMPO studies: Clinical evidence for tavapadone in Parkinson’s disease
The therapeutic benefits of tavapadone were confirmed last year in a series of Phase 3 studies – TEMPO-1, TEMPO-2 and TEMPO-3 2.
In TEMPO-1 and TEMPO-2, tavapadone was studied as monotherapy in people with early-stage Parkinson’s disease (diagnosed less than three years ago). Both studies lasted 27 weeks and used the combined MDS-UPDRS Part II and Part III scores to measure improvements in motor symptoms and daily activities. In TEMPO-1, 529 patients received a fixed daily dose of tavapadone (5 mg or 15 mg). In TEMPO-2, 304 participants received a flexible dose adjusted up to 15 mg per day. In both studies, tavapadone led to significant motor improvements compared to placebo.
TEMPO-3 investigated tavapadone as an adjunctive therapy to levodopa in people with advanced Parkinson’s disease who suffered from motor fluctuations. This study enrolled 507 patients who were taking a stable dose of levodopa but still had at least 2.5 hours of “OFF” time per day. As in TEMPO-2, the researchers used a flexible dosing regimen of 5–15 mg once daily. Participants who took tavapadone in addition to their regular medication experienced approximately one additional hour of good ON time without dyskinesia and a reduction in OFF time of around one hour without an increase in dyskinesia.
Further current studies and classifications can be found in the Parkinson’s blog.
Long-term safety and regulatory outlook for tavapadon
Some participants experienced mild to moderate side effects such as nausea, headaches and dry mouth. Importantly, in the TEMPO-3 study, the frequency of hallucinations and impulse control disorders was comparable to placebo. This suggests that tavapadone avoids many of the side effects associated with D2/D3 receptor stimulation.
A fourth study, TEMPO-4, included 992 participants to investigate the long-term safety and durability of tavapadone’s effects over 58 weeks. The study was completed in December 2025. Although the results are expected shortly, AbbVie has already applied to the FDA for approval to market tavapadone 3. Regulatory review can take up to a year, which means that tavapadone could potentially be approved and launched within that year.
Although Tavapadon does not slow down the progression of the disease, it has the potential to significantly improve the everyday lives of those affected by reducing motor fluctuations and avoiding many of the side effects of existing dopamine therapies. If approved, it could be available to patients relatively soon and enable better symptom control while disease-modifying therapies continue to advance.
Stay tuned for Part 3, where we will focus on cell-based therapies that aim to replace lost dopaminergic neurons and restore brain function. Sign up now for our Parkinson’s newsletter.
More about us
–> Back to the Parkinson’s Blog
References
- Kaye AD, Ford BM, Abbott BM, Broocks KM, Novacic S, Shekoohi S. Emerging Clinical Role of Tavapadon, a Novel Dopamine Partial Agonist, in the Treatment of Parkinson’s Disease. Diseases. 2025;13(9):290. Published 2025 Sep 2. doi:10.3390/diseases13090290
- Bezard E, Gray D, Kozak R, Leoni M, Combs C, Duvvuri S. Rationale and Development of Tavapadon, a D1/D5-Selective Partial Dopamine Agonist for the Treatment of Parkinson’s Disease. CNS Neurol Disord Drug Targets. 2024;23(4):476-487. doi:10.2174/1871527322666230331121028
- https://news.abbvie.com/2025-09-26-AbbVie-Submits-New-Drug-Application-to-U-S-FDA-for-Tavapadon-for-the-Treatment-of-Parkinsons-Disease
