As Parkinson’s disease progresses, the loss of dopamine-producing nerve cells becomes increasingly difficult to control with medication alone. In the last instalment of our three-part series on novel therapies for Parkinson’s disease, we highlight cell-based approaches that aim to replace what the disease destroys. By delivering new dopamine neurons directly into the brain via stem cell therapy, these therapies go beyond merely treating the symptoms and aim to rebuild damaged neural circuits.
How stem cell works in Parkinson’s disease therapy
In 2025, two early-stage clinical trials published in the renowned journals Nature and Cell reported encouraging safety results for stem cell-based therapies in which new dopamine neurons are transplanted directly into the brain. The studies were sponsored by BlueRock Therapeutics (USA, a subsidiary of Bayer AG) and S. Biomedics (Seoul, Korea). They investigated the safety of injecting nerve cells into the brains of people with Parkinson’s disease.
Both clinical trials used dopamine progenitor cells derived from human embryonic stem cells. Although these cells start out as stem cells, they undergo a controlled development process in the laboratory to become so-called “progenitors” – a transitional stage in which they are already committed to developing into dopamine neurons but are not yet fully mature. The use of progenitor cells reduces the risk of uncontrolled growth, which can occur with undifferentiated stem cells. It can be thought of as planting seedlings instead of seeds, which could otherwise grow into unwanted weeds instead of trees.
The main difference between the two studies is that BlueRock Therapeutics uses cryopreserved dopamine progenitor cells called Bemdaneprocel 1, while S. Biomedics uses freshly produced cells called A9-DPCs 2.
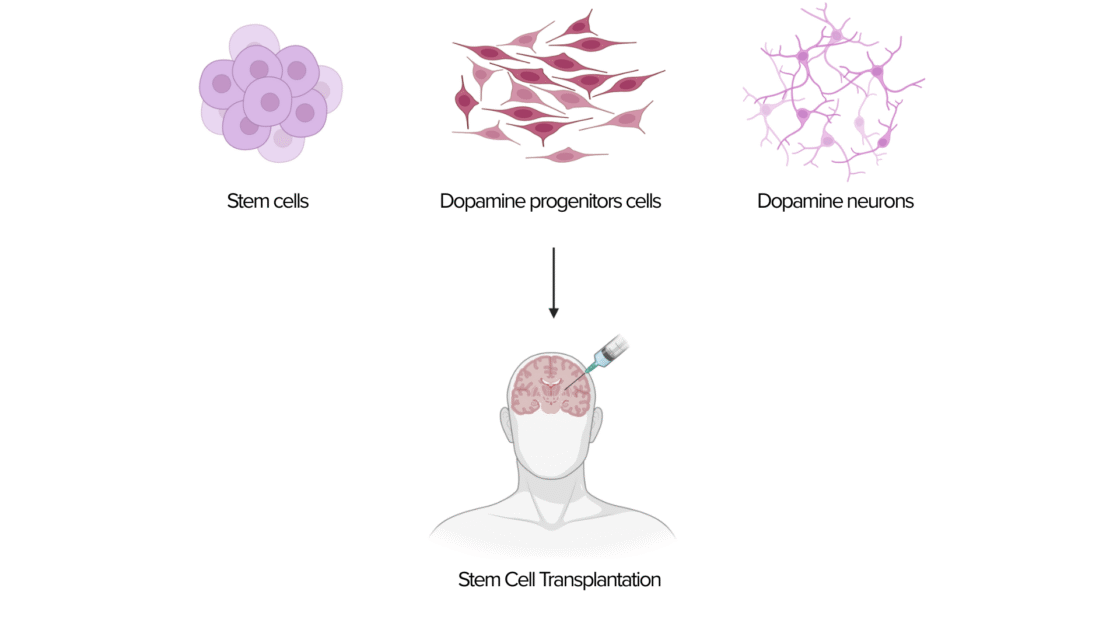
Safety and tolerability of stem cell therapy for Parkinson’s disease
Each study included 12 participants with Parkinson’s disease aged between 50 and 78. Both studies concluded that stem cell therapy was safe and generally well tolerated. Some participants experienced expected side effects related to the surgical procedure (e.g. intracranial haemorrhages) or immunosuppression (e.g. low platelet counts). As with any transplant, participants took immunosuppressive drugs to prevent rejection. Importantly, the transplanted cells did not cause any adverse effects. Imaging studies of the brain showed no signs of tumours, inflammation or infection.
Imaging also confirmed that the transplanted cells survived and developed into functional dopamine neurons. In the Bemdaneprocel study, patients stopped taking immunosuppressive drugs one year after transplantation. Subsequent PET scans six months later showed that the transplanted cells were still alive and functioning – an indication of successful integration into the brain.
Early clinical results: motor function, ON time and medication use
These results could explain the gradual improvements reported by participants in their daily activities and quality of life over a period of 12 to 18 months. In the Bemdaneprocel study, participants reported an average increase of 2.7 hours in daily good ON time. Both studies also showed a reduction in OFF time, which was up to 3.6 hours in the A9-DPC study. Some participants in the A9-DPC study were able to moderately reduce their levodopa dose to avoid medication-induced dyskinesia while their motor function improved.
Cognitive test results remained stable over 12 months in the A9-DPC study, suggesting that neither stem cell therapy nor immunosuppression adversely affected mental clarity. In the Bemdaneprocel study, medication use remained largely unchanged from baseline.
Significance of these findings for the future of Parkinson’s treatment
In 2024, Bemdaneprocel received RMAT (Regenerative Medicine Advanced Therapy) status from the FDA and SAKIGAKE (pioneering regenerative medicine product) approval from Japan’s Ministry of Health, Labour and Welfare. This underscores the great potential of Bemdaneprocel as a possible new therapy for Parkinson’s disease and reflects the confidence of regulatory authorities in its therapeutic benefits. Currently, 102 participants are being recruited for the Phase 3 ExPDite-2 3 study to investigate the effect of stem cell therapy on motor symptoms.
In summary, these studies show that dopamine neuron replacement is moving from theoretical idea to clinical reality. If the results of the Phase 3 trial are positive, stem cell-based therapies could one day provide long-lasting symptom relief and reduce dependence on daily medication.
More about us
–> Back to the Parkinson’s Blog
References
- Tabar V, Sarva H, Lozano AM, et al. Phase I trial of hES cell-derived dopaminergic neurons for Parkinson’s disease. Nature. 2025;641(8064):978-983. doi:10.1038/s41586-025-08845-y
- Chang JW, Na HK, Chang KW, et al. Phase 1/2a clinical trial of hESC-derived dopamine progenitors in Parkinson’s disease. Cell. 2025;188(25):7036-7048.e11. doi:10.1016/j.cell.2025.09.010
- https://clinicaltrials.gov/study/NCT06944522
